- Short description
- Low-grade inflammation causes gap junction-coupled cell dysfunction throughout the body, which can lead to the spread of systemic inflammation
- Anti-inflammatory effects induced by pharmaceutical substances on inflammatory active brain astrocytes
- Influence of inflammation on actin cytoskeleton on oral keratinocytes
- Therapeutic innovation: networks of inflammatory-reactive chondrocytes as targets of inflammation
- Research partners
The Glial Cell Research Group
Short description
Network coupled cells, the supporting cells in different organs in the body, such as astrocytes in the nervous system and chondrocytes in the joints, regulate their cellular homeostasis via calcium signals spread between the cells through gap junctions. These networks can be affected by inflammatory stimuli and become dysregulated. It can give rise to systemic inflammation, which in turn can lead to pain that can turn into chronic. Our results with different combinations of pharmaceuticals, will lead to clinical implications. Our purpose is now to continue with pre-clinical and clinical trials in vivo.
Low-grade inflammation causes gap junction-coupled cell dysfunction throughout the body, which can lead to the spread of systemic inflammation
Inflammation is a physiological response to injury that is designed to remove dangerous stimuli. Low-grade inflammation can be initiated in vivo after traumatic injury or in chronic diseases such as neurodegenerative, metabolic and autoimmune diseases (Abbott et al, 2006). Connexin-linked gap junction-coupled cell networks can be targeted, leading to the spread of inflammation and changes in biochemical cellular parameters (Hansson & Skiöldebrand, 2015). It can affect surrounding cells that can cause spread of inflammatory substances, thus affecting other network-linked cells in other organs in the body, which can cause systemic inflammation.
Several organs in the body comprise cells coupled into networks that communicate with each other through gap junctions. Examples of such cellular networks are astrocytes in the brain, keratinocytes in the skin and buccal membranes, epithelial cells in several organs, connective tissue cells such as chondrocytes in the articular cartilage and tenocytes in ligaments and cardiac myofibroblasts in the heart (Hansson & Skiöldebrand, 2019).
Astrocytes in the CNS are the most well studied network-coupled cells that play a pivotal role in chronic neuroinflammation (Hansson, 2010). Inducers of inflammation trigger the production of inflammatory mediators, which alter the functionality of tissues and organs and lead to harmful changes in different barrier systems (Rönnbäck & Hansson, 2017; 2019).
Anti-inflammatory effects induced by pharmaceutical substances on inflammatory active brain astrocytes
Background
Pharmaceutical treatment with probable anti-inflammatory substances that attack cells in various ways including receptors, ion channels, or transporter systems may slow down the progression of inflammatory conditions. Network coupled cells, such as astrocytes, regulate their cellular homeostasis via Ca2+ signals spread between the cells through gap junctions. Intracellular Ca2+ release is controlled by different signaling pathways that can be stimulated by ATP, glutamate and 5-HT. Astrocytes and microglia are the most prominent target cells for inflammation in the CNS. Their responses upon inflammatory stimuli work through the NO/cyclic GMP/protein kinase G systems that can downregulate the ATP-induced Ca2+ signaling, as well as G protein activities which alter Na+ transporters including the Na+/K+-ATPase pump activity, Toll-like receptor 4 (TLR4), glutamate-induced Ca2+ signaling, and release of pro-inflammatory cytokines.
Aims
The rationale for the project is to investigate a combination of pharmaceutical substances influencing the NO and the Gi/Gs activations of inflammatory reactive cells in order to make the cells return into a more physiological state. The ATP-evoked Ca2+ signaling is important, maybe due to increased ATP release and subsequent activation of purinergic receptors. A balance between intercellular Ca2+ signaling through gap junctions and extracellular signaling mediated by extracellular ATP may be important for physiological functions.
Results
Combinations of different pharmaceutical substances downregulated 5-HT-, glutamate- and ATP-evoked intracellular Ca2+ release to a more normal Ca2+ signaling state. Furthermore, inflammatory receptor expressions decreased and the Na+/K+-ATPase pump activity increased (Forshammar et al, 2011; Block et al, 2012; 2013 a, b; 2015; Skiöldebrand et al, 2016; Nunes et al, 2016; Hansson et al, 2018; Hansson & Skiöldebrand, 2019).
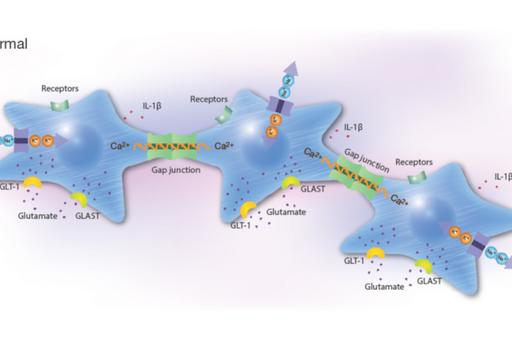
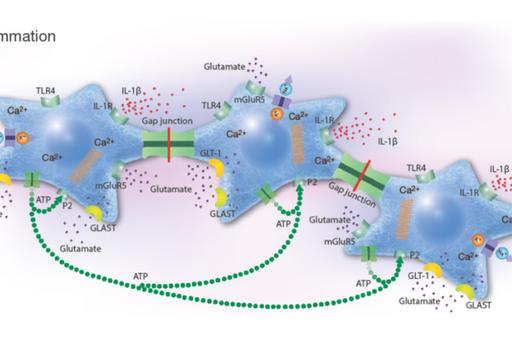
Conclusions
Our unique results suggest that a combination of pharmaceuticals, especially at the right concentrations that can be extremely low, is necessary to restore vital cellular functions in astrocytes. Thus, inflammatory processes could be inhibited or restored to normal physiological homeostasis. Our purpose is now to continue with pre-clinical and clinical trials in vivo.
Influence of inflammation on actin cytoskeleton on oral keratinocytes
Background
Several oral mucosal diseases are associated with inflammation where environmental factors such as nicotine in tobacco and oral bacteria can be of importance (Hansson & Skiöldebrand, 2019). The keratinocytes seem to play a major role. The actin cytoskeleton and the TLRs are affected significantly. The actin cytoskeleton is a key regulator of ion transport in cells and is a dynamic structure that regularly undergoes remodeling. The cytoskeleton integrity and Ca2+ waves has been assessed as indicators of inflammatory state. In wound healing the keratinocytes are active in the cytoskeleton dynamics. The oral mucosa is composed of stratified squamous epithelial tissue, mainly keratinocytes. The cells are attached to a basement membrane which separates the mucosa from connective tissue. Blood vessels and nerves are restricted to the connective tissue; therefore, nutrients to the cells have to diffuse into the cell-layers. The keratinocytes exhibit numerous cytoplasmic processes attached to adjacent keratinocytes by desmosomes, adherens junctions, tight junctions and gap junctions.
Aims
The purposes are to evaluate a brushing method and to test future possibilities of using it for analysis in an inflamed tissue or chronic pain conditions. Keratinocytes are obtained from the buccal mucosa. The cells are histologically studied and evaluated in changes of the actin cytoskeleton and in expression of different biochemical parameters. Different oral mucosal diseases are studied in comparison to healthy controls.
Results
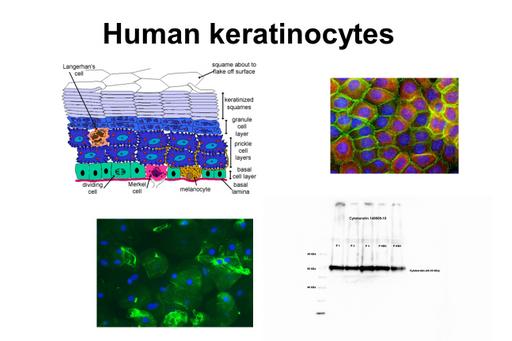
The results show a clear identification of keratinocytes stained with histochemistry and immunocytochemistry and cytokeratin being identified with Western blot. Analysis and comparison between healthy patients and patients with lesions showed a statistically significant difference between the integrated density of TLR4 in patients with oral lichen planus (OLP) and of β-actin in patients with leukoplakia. This was noticed as a higher integrated density of TLR4 and a lower integrated density of β-actin, respectively.
Conclusions
The present study shows that keratinocytes and proteins are present in the obtained samples validating the brushing method and making it an acceptable method to study changes in an inflamed tissue. Examination work 2019 by Marina Baza, Department of Oral Medicine and Pathology, Institute of Odontology, The Sahlgrenska Academy, University of Gothenburg, Sweden.
Therapeutic innovation: networks of inflammatory-reactive chondrocytes as targets of inflammation
Background
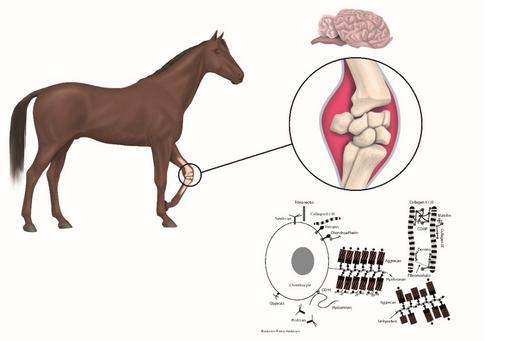
Osteoarthritis (OA) is a low-grad inflammatory disease affecting one or several joints and are leading to severe pain and disability in humans and animals. Substantial progress has been made in understanding how chronic systemic low-grade inflammation influences the physiology of several diseases but not why it fails to resolve in OA, metabolic syndromes, and autoimmune diseases. The physiological nature of inflammation has been gaining ground as a key contributor and search for inflammatory markers has increased. Most of OA research are based on induced (biochemically and traumatically) OA in laboratory animals that not develop OA spontaneously (mice and rats). To our knowledge the disease has to be studied in research animals (both in vitro and in vivo) that develop the disease spontaneously and where the early events can be characterised.
Aims
With a high-throughput screening system test new and better combinations of pharmaceutical compounds targeting dysregulated cellular mechanisms in chondrocytes. The screening system has high reproducibility measuring intra- and extracellular Ca2+ in living cells and enables us to test the efficacy of pharmaceutical compounds and combinations thereof on a large scale. We will prevent chondrocytes from becoming inflammatory-reactive and/or restore chondrocytes, which are inflammatory-reactive and rescue them back to physiological homeostasis regarding the analysed parameters.
Results
Chondrocytes showed different metabolic properties according to the joint status of the original tissue (healthy vs OA). The results indicate that chondrocytes from OA joints are predisposed to show an exaggerated response to inflammatory stimuli, such as increased expression of pain mediators, which is absent in chondrocytes of healthy joints.
Biochemical alterations in inflammatory reactive chondrocytes suggesting evidence for intercellular network communication between chondrocytes is observed in vitro (Skiöldebrand et al, 2018). Furthermore, serotonin-evoked cytosolic Ca2+ release and opioid receptor expression are upregulated in articular cartilage chondrocytes from osteoarthritic joints in horses (Skiöldebrand et al, 2019).
A combination of three different pharmacological substances in ultra-low concentrations have shown to restore inflamed chondrocytes coupled in networks in vitro (Hansson & Skiöldebrand, unpublished). We have a patent registered for the drug combination and have tested the drug combination in some clinical cases, horses with OA. Results showed a decline in COMP fragment concentration in synovial fluid and serum and reduction in clinical symptoms (lameness) after treatment, which indicate that the drug combination has disease modifying properties which is unique.
Conclusions
According to the present study, drug combinations at the right concentrations, especially extremely low concentrations, affect different cellular biochemical mechanisms and represent a potential solution for downregulating inflammatory parameters, thereby restoring cells or networks to normal physiological homeostasis. In vivo reduction in lameness in horses with OA is seen. Our purpose is now to continue with pre-clinical and clinical trials in vivo.
Research partners
Bengt Hasséus, University of Gothenburg
Eva Skiöldebrand, Swedish University of Agricultural Sciences
Anders Lindahl, University of Gothenburg
Catarina Raposo, University of Campinas
Linda Block, University of Gothenburg
Jairo Robledo-Sierra, University CES


